Value
CLINICAL PROBLEM
Rotator Cuff Disease (see Figure 1) is the most common shoulder disorder in America. Approximately one- quarter of adults over 40 years of age in the United States, and more than half of adults over the age of 60, have a rotator cuff tear. There are an estimated 4 million people with Rotator Cuff Disease who are at risk for disability. Though some people experience acute tears of the rotator cuff via injury, this is primarily a degenerative disease; the older the patient population, the more prevalent the disorder becomes.

To complicate matters, patients that undergo surgical repair of the rotator cuff do not experience consistent, lasting results. There is a veritable cornucopia of surgical approaches, repair configurations, and implants and instrumentation that are available to surgeons based upon the anatomy and pathology of the patient’s rotator cuff tear; nevertheless, 30-90% of repaired rotator cuffs (accounting for variations in technique, equipment, disease progression, and skill) experience a subsequent tear (2).

Surgically repaired rotator cuffs fail in two predominant locations – at the site of surgical re-attachment of tendon to bone, and in the tendon medial to the re-attachment site (Figure 2). If the re-tear occurs at the medial tendon, it is commonly accepted to be a result of inadequate tendon thickness. This has been a focus of several recent innovations, such as Regeneten (Smith & Nephew), BioBrace (ConMed), and Tapestry RC (Zimmer Biomet), which aim to reinforce and increase the thickness of the medial tendon.
However, these products may not address the site of surgical re-attachment of tendon to bone. In native, healthy rotator cuffs – as well as every other hard-to-soft tissue junction in the human body – an enthesis is present. The enthesis is a thin, transitory region that exhibits characteristics of both bone and tendon and plays a vital role in vascularization and load distribution of the tendon to bone junction.
When a rotator cuff is surgically repaired, it is common for the body to restore the mechanical connection with scar tissue instead of a new enthesis, thereby critically compromising the short-term healing potential and long-term stability of the surgically repaired rotator cuff.

It is clear that patients need additional advancements and technologies that seek to decrease the rates of rotator cuff re-tears by assisting the critical restoration that must occur at the bone to tendon junction to set the foundation for a long-lasting rotator cuff repair that restores function and mobility.
SOLUTION
BioEnthesis®

biphasic interpositional
allograFt FOR rotator cuff repair
BioEnthesis is a biphasic allograft composed of human cancellous bone. It is designed for interpositional implantation to provide an interconnective porous matrix that retains biofluids expressed from humeral surface and reflects the native anatomical transition from soft to rigid tissue present in the enthesis. BioEnthesis consists of demineralized, highly porous collagenous bone that may support soft tissue integration at the tendon-graft interface and a mineralized, osteointegrative, cancellous contact layer at the bone-graft interface all within a single allograft embodiment.
EVIDENTIARY SUPPORT
1: Existing Literature - Importance of the Enthesis
“At the rotator cuff, repair techniques focus on secure attachment of tendon to bone using sutures and bone anchors, but have not been effective in promoting regeneration of the native enthesis structure. Therefore, the mechanical properties of the fibrovascular scar tissue that forms at the interface do not approximate those of the normal tendon enthesis.” (4)
2: Proof of Concept in Ovine Model
“Scaffold-repaired tendons displayed histological evidence of a transitional zone from tendon to fibrocartilage to bone. The transition was completely absent in tendons treated with standard primary repair. Instead, disorganized scar tissue filled the defect area between the tendon and the bone.”
“The material produced a four-zone fibrocartilaginous interface similar to the normal interface structure.”

3: Ovine Study
4. Completed Cases
105 cases completed with zero adverse events reported and positive PRO and MRI results. See an exemplary patient below:
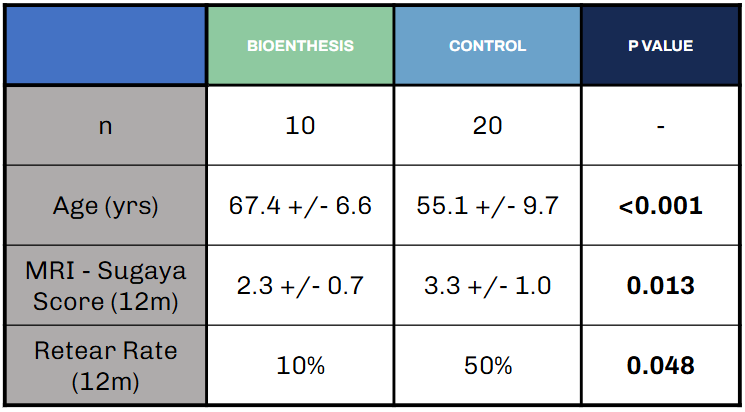
5. Clinical Study
An investigator-initiated study was completed at Midwest Orthopedics at Rush evaluating BioEnthesis in rotator-cuff procedures*. Preliminary 12-month observations included assessments of structural outcomes (e.g., Sugaya classification) and re-tear rates in the study cohort, including patients with differing baseline characteristics
*The study was conducted under institutional IRB approval and is supported by an unrestricted educational grant, with no sponsor involvement in study design, conduct, or data analysis.
REFERENCES
1. “Rotator Cuff Injury.” Mayo Clinic, Mayo Foundation for Medical Education and Research, 11 May 2023, www.mayoclinic.org/diseases-conditions/rotator-cuff-injury/symptoms-causes/syc-20350225.
2. Dickerson, Darryl A et al. “In vitro and in vivo evaluation of orthopedic interface repair using a tissue scaffold with a continuous hard tissue-soft tissue transition.” Journal of orthopaedic surgery and research vol. 8 18. 19 Jun. 2013, doi:10.1186/1749-799X-8-18
3. Bianchi E, Ruggeri M, Rossi S, Vigani B, Miele D, Bonferoni MC, Sandri G, Ferrari F. Innovative Strategies in Tendon Tissue Engineering. Pharmaceutics. 2021 Jan 11;13(1):89. doi: 10.3390/pharmaceutics13010089. PMID: 33440840; PMCID: PMC7827834.
4. Derwin, Kathleen A et al. “Enthesis Repair: Challenges and Opportunities for Effective Tendon-to-Bone Healing.” The Journal of bone and joint surgery. American volume vol. 100,16 (2018): e109. doi:10.2106/JBJS.18.00200
5. National average physician fee schedule via CMS.gov.
6. National average outpatient facility payment via CMS.gov.







